 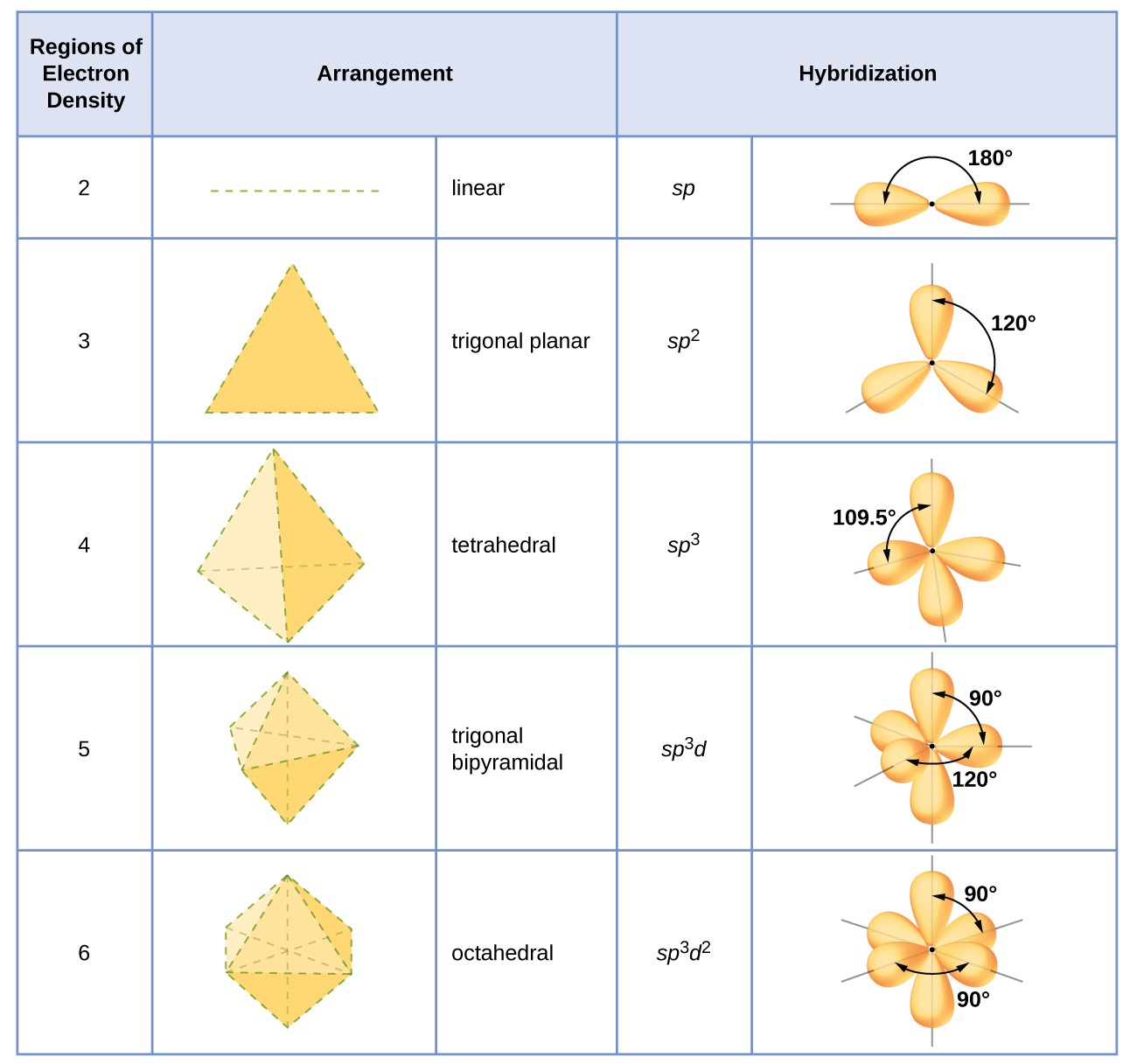 
Sp 3d 2 hybridization involves atomic orbitals of same electron shell.ĭ 2sp 3 hybridization involves atomic orbitals of two electron shells. Sp 3d 2 hybridization forms sp 3d 2hybrid orbitals.ĭ 2sp 3 hybridization d 2sp 3 hybrid orbitals. Sp 3d 2 hybridization is the mixing of s, p and d atomic orbitals of the same electron shell to form sp 3d 2 hybrid orbitals.ĭ 2sp 3 hybridization is the mixing of s and p atomic orbitals of the same electron shell with d orbitals of another electron shell to form d 2sp 3 hybrid orbitals. Ex: SF6 molecule has an octahedral shape because the 3s, 3p and 3d atomic orbitals of the sulfur atom (S) are mixed to formsp 3d 2 hybrid orbitals. Let us consider an example in order to understand the sp 3d 2 hybridization. The octahedral arrangement displays a square plane having four hybrid orbitals and the two remaining orbitals are oriented above and below of this square plane (perpendicular to this plane). These hybrid orbitals have 90 o angles between two orbitals in the octahedral arrangement. The sp 3d 2 hybrid orbitals are arranged in octahedral arrangement. This mixing results in six hybrid orbitals of same size and shape but different from their orientation. There, one s atomic orbital, three p atomic orbitals and two d atomic orbitals mix with each other. 
Sp 3d 2 hybridization is the mixing of s, p and d atomic orbitals of the same electron shell to form sp 3d 2 hybrid orbitals. Side by Side Comparison – sp3d2 vs d2sp3 Hybridization in Tabular Form Similarities Between sp3d2 and d2sp3 Hybridizationĥ. The key difference between sp 3d 2 and d 2sp 3hybridization is that sp 3d 2 hybridization involves atomic orbitals of same electron shell whereas d 2sp 3 hybridization involves atomic orbitals of two electron shells. sp 3d 2 and d 2sp 3 are such hybrid orbitals. Hybridization is the mixing of atomic orbitals to form hybrid orbitals. Hybridization of orbitals occurs in order to obtain suitable shapes required for chemical bonding. Atomic orbitals can undergo a process known as hybridization. Different scientific discoveries have proposed different shapes for these orbitals. There are hypothetical structures known as orbitals in an atom in which electrons reside. (2012), Ch.Key Difference – sp3d2 vs d2sp3 Hybridization John McMurry, Organic Chemistry 8th ed.Other atoms such as nitrogen, phosphorus, oxygen, and sulfur also use hybrid orbitals to form strong, oriented bonds. When forming a triple bond with linear geometry, carbon uses two equivalent sp hybrid orbitals and two unhybridized p orbitals. When forming a double bond with planar geometry, carbon uses three equivalent sp 2 hybrid orbitals and one unhybridized p orbital. When forming only single bonds with tetrahedral geometry, carbon uses four equivalent sp 3 hybrid orbitals. In the valence bond description, carbon uses hybrid orbitals to form bonds in organic molecules.In chemistry, hybridisation (or hybridization) is the concept of mixing atomic orbitals into new hybrid orbitals (with different energies, shapes, etc., than the component atomic orbitals) suitable for the pairing of electrons to form chemical bonds in valence bond theory. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
